Phage Therapy
Nature's evolving solution to bacterial infections
As bacterial resistance to antibiotics grows exponentially and new antibiotic discovery declines, bacteriophages have re-emerged as highly promising alternatives for tackling this crisis.
The Silent Pandemic
Antimicrobial resistance (AMR) is a critical global health challenge and is ranked by the World Health Organization (WHO) among the top ten threats to public health worldwide. Often described as a “silent pandemic,” AMR is responsible for an estimated 1.3 million direct deaths each year and contributes to nearly 4.95 million deaths overall. Without effective intervention, annual direct deaths are projected to rise to as many as 10 million by 2050. In Europe alone, approximately 250,000 patients suffer from complex, difficult-to-treat infections, resulting in more than 35,000 deaths annually.
As bacterial resistance to antibiotics grows exponentially and new antibiotic discovery declines, bacteriophages have re-emerged as a highly promising alternative for tackling this crisis.
What is a bacteriophage?
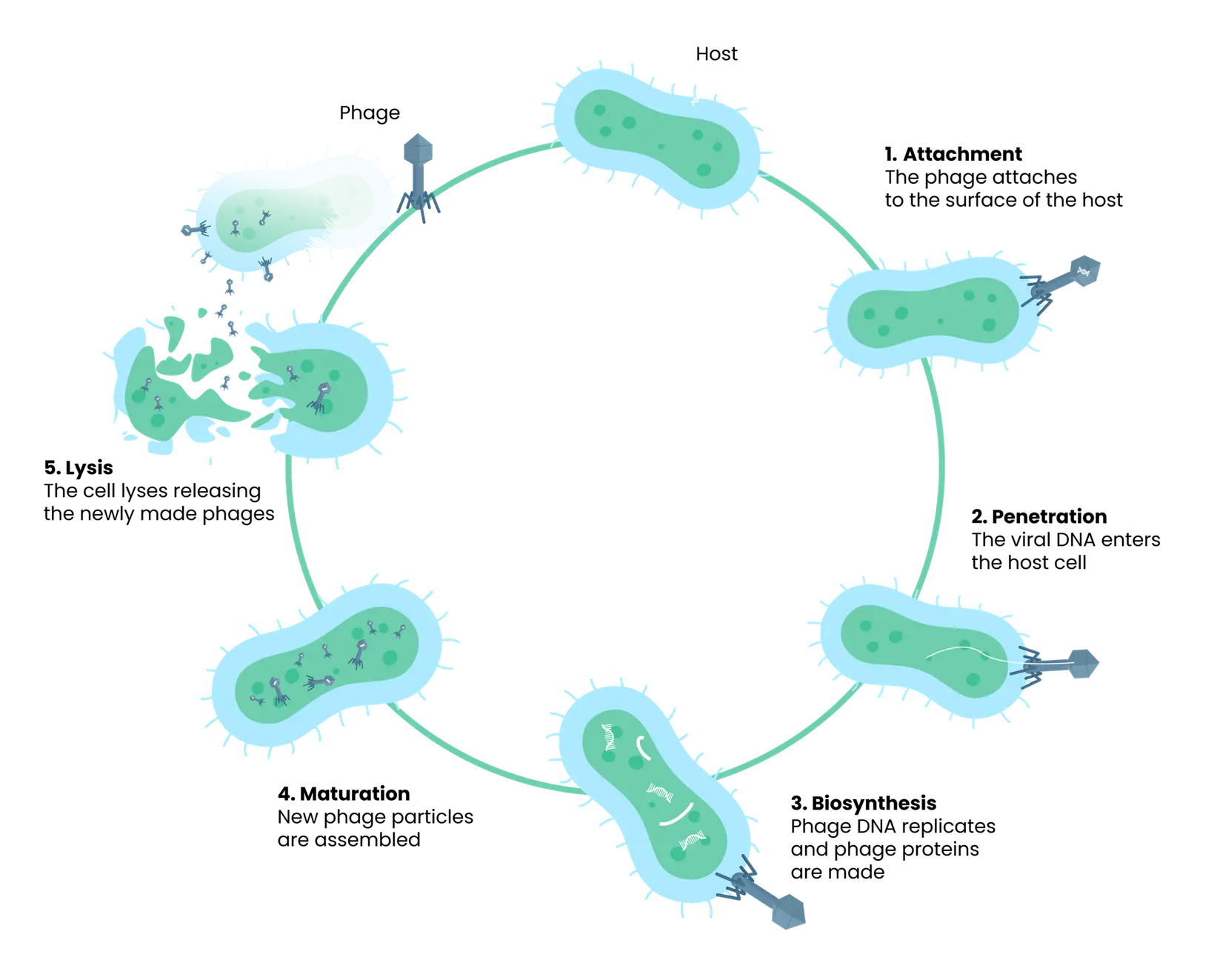
A bacteriophage, or phage, is a naturally occurring virus that specifically and exclusively infects bacteria. Phages recognize and attach to particular receptors on bacterial cells, inject their genetic material, and hijack the bacterium’s internal machinery to produce new phages. This process ultimately leads to the destruction of the target bacterium, while leaving commensal or beneficial bacteria along with human cells unaffected, which underpins their potential as a targeted therapeutic option against bacterial infections.
The phage lifecycle consists of a series of well-defined steps, from bacterial recognition and entry to replication and release of new phages.
The infographic below illustrates the key stages of the bacteriophage lifecycle.
From Phage Discovery to Clinical Implementation
Because bacteriophages are highly specific to their bacterial targets, identifying the right phage for each pathogen is a critical step in phage therapy. Our scientists and partners continuously search for new bacteriophages in environments where bacteria naturally thrive. From clinical samples and human-associated water systems to selected natural environments, bacteriophages can be found in a wide range of settings characterized by high microbial diversity.
Each newly identified bacteriophage undergoes a rigorous screening and characterization process by our R&D team. We only keep phages that meet strict quality and safety criteria and show strong activity against the bacteria we focus on, and then add them to our curated phage collection.
Once selected, our production team develops scalable manufacturing and release workflows for bacteriophages, aligned with pharmaceutical-quality expectations. In parallel, we are exploring hospital pharmacy supply models designed to enable access to quality-controlled phage candidates through applicable clinical and regulatory pathways.
In parallel, our Phagogram team is developing PhageXpert™, our proprietary phagogram platform, and works to integrate selected phages into the assay panel designed to support rapid testing of patient-derived bacterial isolates in hospital-lab workflows.
Finally, the phage activity profile can be reviewed by the hospital team to support treatment planning, where appropriate and in line with applicable clinical and regulatory pathways.

Manufacturing and supply capabilities described are under development and are not yet authorised for clinical use or commercial supply in the EU. This content is provided for scientific and corporate information purposes only and is not an offer to supply phage products for patient administration.
Phage Therapy
Phage therapy is not a one-size-fits-all approach. Because phages are often specific to particular bacteria, treatment strategies may differ from one infection to another. Many in the field support a more personalised approach, while also recognising the value of greater standardisation, especially around clinical protocols that are tailored to different indications.
A key practical consideration is delivering active phages to the site of infection, and the most suitable route of administration often depends on the infection’s location. Reported approaches include topical use for skin and burn infections, local delivery in some bone and joint settings, nebulisation for respiratory infections and other methods adapted to the clinical context.
Our Areas of Focus
As clinical experience with phage therapy grows, clinical teams are increasingly sharing knowledge and treatment protocols. This collective learning has produced encouraging results in several indications, while also suggesting that outcomes may be more favourable in some infection types than in others.
These insights guide our work. Drawing on emerging evidence, medical urgency and practical experience, Inteliphage initially focuses on indications where phage therapy has been reported most consistently and where the clinical need is highest.

Bone & Joint Infections

Respiratory Tract Infections

Urinary Tract Infections

Bloodstream Infections

Skin & Soft Tissue Infections
And Antibiotics?
Phage therapy is generally discussed as a complement to antibiotics, not a replacement. In practice, it may be used alongside standard treatment depending on the infection, the patient and local protocols. Some studies and clinical reports suggest that certain phage–antibiotic combinations can work well together in specific settings.
Both antibiotics and phages can face resistance over time. Because phages and bacteria can adapt to each other, researchers are exploring how these dynamics, and possible resistance trade-offs, might be used to support treatment strategies. This remains an active area of research and outcomes can vary by infection and context.