One Platform for Phage Therapy
One of the biggest barriers to routine clinical use of phage therapy is operational complexity: limited access to expertise, regulatory constraints and end-to-end logistics. From identifying the right protocol and diagnostic testing partner to shipping isolates and sourcing suitable phages, the operational burden can delay clinical decision-making.
Inteliphage aims to bring the key tools for effective phage therapy into the hospital enabling faster decisions with greater confidence.
The Inteliphage Solution
From pure patient sample to active phage selection in hours
From active phage to personalized formulation in minutes
The Inteliphage Solution
From pure patient sample to active phage selection in hours
From active phage to personalized formulation in minutes
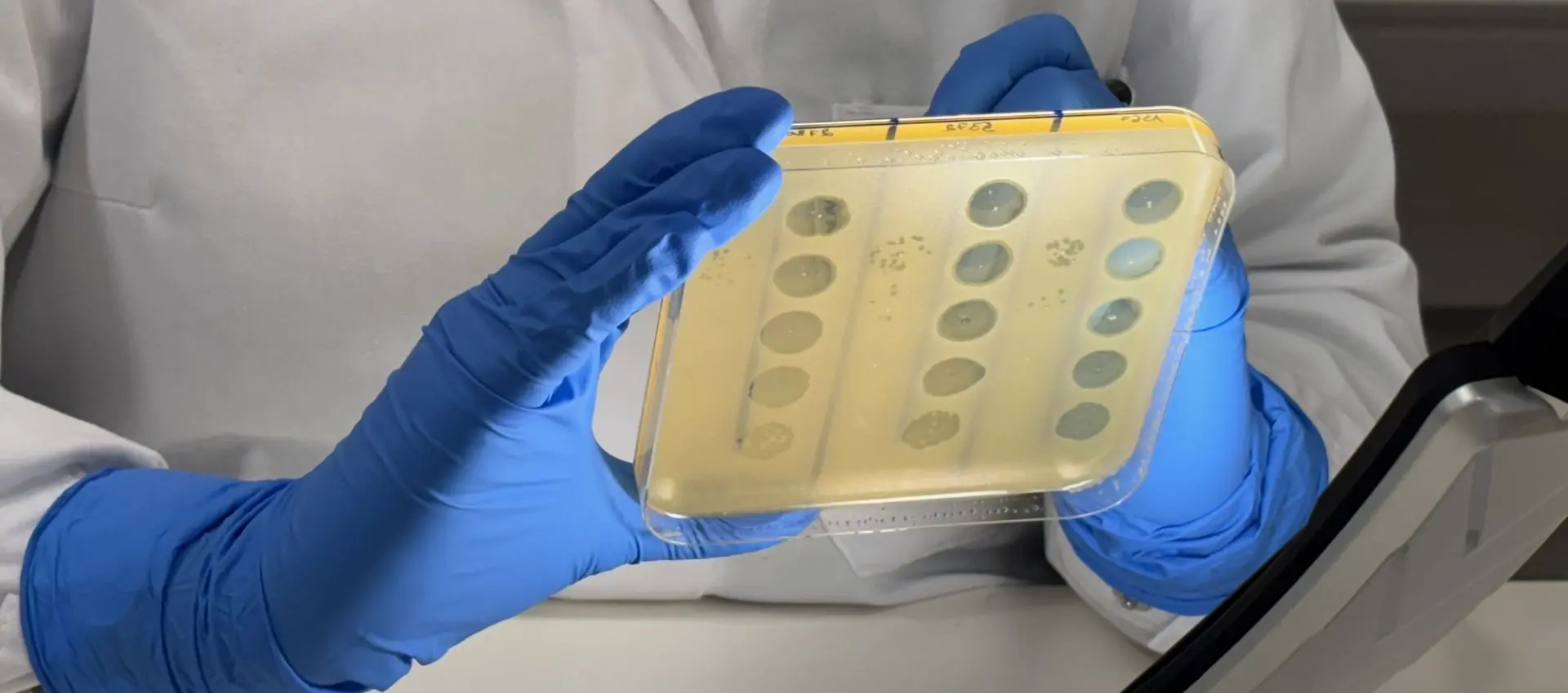
Effective phage therapy starts with identifying phages that match a patient’s bacterial isolate. Inteliphage is developing PhageXpert™, a patented and easy-to-use phagogram workflow designed for hospital-lab operations.
PhageXpert™ is being developed to support parallel testing of multiple phages on a patient isolate within hours. Using single-use kits and an automated workflow, it is designed to simplify handling and reduce operator variability, minimizing the need for specialized phage expertise.
The system is being developed to generate a qualitative phage-activity profile based on the susceptibility of a patient isolate, standardising how phage activity is measured and reported.
From sample to result in approx. 4 hours
Possibility to screen up to 20 phages in 1 run
Ensuring effective phage selection, improving patient outcome
PhageXpert™ is being developed as an in vitro diagnostic device under the EU IVDR. It is not CE-marked and is not yet available for clinical use in the EU.
Inteliphage is building an integrated in-hospital workflow that links rapid phage-activity profiling with access to quality-controlled phage candidates through applicable clinical and regulatory pathways, streamlining the route from sample to treatment planning.
Phage Library
Inteliphage maintains an extensive phage bank, continuously expanded and characterised to support the selection of high-performing phage candidates for further development and quality-controlled manufacturing.
In parallel, we are establishing GMP-ready manufacturing and release processes and exploring hospital pharmacy supply models to enable access to quality-controlled phage candidates through applicable clinical and regulatory pathways. Where appropriate, this approach is designed to support timely treatment planning for difficult-to-treat infections.
Our collection mainly targets ESKAPE+ pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter spp.,).
Large Phage Bank targetting most ESKAPE pathogens
Produced with highest quality standards ensuring sterility & phage purity
Continuously improving host ranges to stay adaptable & relevant to geographical needs
References to phage banking, GMP-ready manufacturing and hospital pharmacy supply models describe development activities and do not imply authorisation for clinical use or commercial supply in the EU. Any patient use, where permitted, would occur only through applicable clinical and regulatory pathways.

Alaeddine Meghraoui
Clinical Implementation Scientist

Gunther Vanwezer
Chief Business Officer

Steven De Soir
Product Manager